 The first step of AAV infection often requires the attachment of the capsid to carbohydrate moieties on the cell surface. Of the 11 well-characterized AAV serotypes, heparan sulfate proteoglycan and sialic acid have been suggested to be the attachment receptors for AAV type 2 and types 4 and 5, respectively. In this report, we identify the receptor for the two closely related serotypes, AAV1 and AAV6. First, we demonstrate using coinfection experiments and luciferase reporter analysis that AAV1 and AAV6 compete for similar receptors. Mucin competition was carried out by pretreating rAAV with bovine submandibular gland mucin (Sigma) for 30 min at 20°C. AAV vectors, in the presence or absence of mucin, were added to Cos-7 cells in equal volumes of DMEM for 1 h at 37°C. Cells were rinsed twice with DMEM and incubated at 37°C. Two thousand particles/cell were pretreated with mucin (concentrations up to 0.5 mg/ml), and cells were assayed for luciferase expression 24 h posttransduction. After incubation for 1 h at 37°C, cells were transduced at a multiplicity of infection (MOI) of 2 × 103 for 1 h with virus, and luciferase expression was analyzed 24 h after transduction. Unlike heparin sulfate, enzymatic or genetic removal of sialic acid markedly reduced AAV1 and AAV6 binding and transduction.
The first step of AAV infection often requires the attachment of the capsid to carbohydrate moieties on the cell surface. Of the 11 well-characterized AAV serotypes, heparan sulfate proteoglycan and sialic acid have been suggested to be the attachment receptors for AAV type 2 and types 4 and 5, respectively. In this report, we identify the receptor for the two closely related serotypes, AAV1 and AAV6. First, we demonstrate using coinfection experiments and luciferase reporter analysis that AAV1 and AAV6 compete for similar receptors. Mucin competition was carried out by pretreating rAAV with bovine submandibular gland mucin (Sigma) for 30 min at 20°C. AAV vectors, in the presence or absence of mucin, were added to Cos-7 cells in equal volumes of DMEM for 1 h at 37°C. Cells were rinsed twice with DMEM and incubated at 37°C. Two thousand particles/cell were pretreated with mucin (concentrations up to 0.5 mg/ml), and cells were assayed for luciferase expression 24 h posttransduction. After incubation for 1 h at 37°C, cells were transduced at a multiplicity of infection (MOI) of 2 × 103 for 1 h with virus, and luciferase expression was analyzed 24 h after transduction. Unlike heparin sulfate, enzymatic or genetic removal of sialic acid markedly reduced AAV1 and AAV6 binding and transduction.
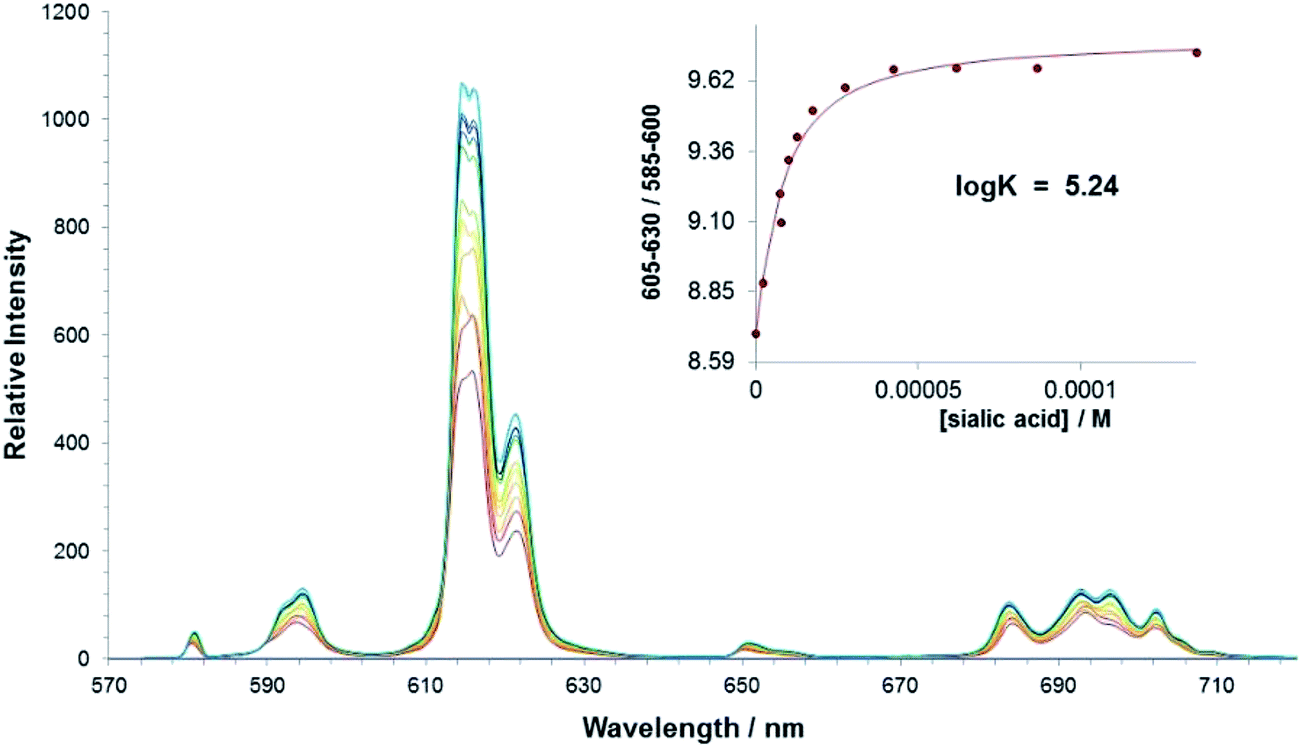 We used cell-based assays to show that α2,3 and α2,6 sialic acids that are present on N-linked glycoproteins facilitate cellular transduction by both AAV1 and AAV6 vectors. Taken together, competition, genetic, inhibitor, enzymatic reconstitution, and glycan array experiments support α2,3 and α2,6 sialic acids that are present on N-linked glycoproteins as primary receptors for efficient AAV1 and AAV6 viral infection. Furthermore, a resialylation experiment on a deficient Lec-2 cell line confirmed a 2,3 and 2,6 N-linked sialic acid requirement, while studies of mucin with O-linked sialic acid showed no inhibition effect for AAV1 and AAV6 transduction on Cos-7 cells. The cell suspension was filtered and exposed to red blood cell lysis as described above. The relative expression for each gene was calculated according to the 2−ΔΔCt method described by Livak and Schmittgen.22 The efficiency of the amplification reaction for each primer/probe is above 95% (as determined by the manufacturer). Recombinant adeno-associated viruses (AAVs) are promising vectors in the field of gene therapy. Adeno-associated viruses (AAVs), dependoviruses of the parvovirus family, rely on a helper virus, such as adenovirus or herpesvirus, to complete their life cycle. Interestingly, an older version of the array, containing glycans immobilized as biotinylated glycosides on a 384-well streptavidin-coated plate, was used to screen the sugar binding specificity of the parvovirus minute virus of mice (MVM) capsids (M.
We used cell-based assays to show that α2,3 and α2,6 sialic acids that are present on N-linked glycoproteins facilitate cellular transduction by both AAV1 and AAV6 vectors. Taken together, competition, genetic, inhibitor, enzymatic reconstitution, and glycan array experiments support α2,3 and α2,6 sialic acids that are present on N-linked glycoproteins as primary receptors for efficient AAV1 and AAV6 viral infection. Furthermore, a resialylation experiment on a deficient Lec-2 cell line confirmed a 2,3 and 2,6 N-linked sialic acid requirement, while studies of mucin with O-linked sialic acid showed no inhibition effect for AAV1 and AAV6 transduction on Cos-7 cells. The cell suspension was filtered and exposed to red blood cell lysis as described above. The relative expression for each gene was calculated according to the 2−ΔΔCt method described by Livak and Schmittgen.22 The efficiency of the amplification reaction for each primer/probe is above 95% (as determined by the manufacturer). Recombinant adeno-associated viruses (AAVs) are promising vectors in the field of gene therapy. Adeno-associated viruses (AAVs), dependoviruses of the parvovirus family, rely on a helper virus, such as adenovirus or herpesvirus, to complete their life cycle. Interestingly, an older version of the array, containing glycans immobilized as biotinylated glycosides on a 384-well streptavidin-coated plate, was used to screen the sugar binding specificity of the parvovirus minute virus of mice (MVM) capsids (M.
In the present study, we focused on determining the linkage specificity of sialic acid binding for AAV1 and AAV6 transduction. In contrast, resialylation with α2,3(O)-sialyltransferase did not affect AAV1 and AAV6 transduction, while resialylation with α2,3(N)- or α2,6(N)-sialyltransferase resulted in substantial increases in AAV1 and AAV6 transduction (Fig. (Fig. If you have any inquiries regarding where and how you can use sialic acid manufacturers, you can contact us at our own webpage. 8A).8A). AAV6 and newly identified type 6-like variants also required a form of sialic acid for productive infection (35). However, Seiler et al. In addition, AAV, like most viruses, requires the presence of coreceptors for productive infection. In order to improve client engagement and boost sales, market participants are now concentrating on building their digital presence and reputations. What focused approach and constraints are holding the Sialic Acid market? Further analysis using lectin staining and lectin competition assays identified that AAV1 and AAV6 use either α2,3-linked or α2,6-linked sialic acid when transducing numerous cell types (HepG2, Pro-5, and Cos-7). For example, a single extrachromosomal vector can include multiple expression cassettes or more that one compatible extrachromosomal vector can be used maintain an expression cassette in a host cell.
Lectin competition experiments were done by preincubating cells with 100 μg/ml of either WGA, MAA, or SNA (Vector Laboratories Inc.) in medium at 4°C for 10 min. The cells were treated with 50 mU/ml neuraminidase type III from Vibrio cholerae (Sigma, St. Louis, Mo.) at 37°C for 2 h or were untreated and then chilled at 4°C for 15 min. Resialylation was carried out with 50 mU/ml sialyltransferase in α-MEM for 2 h at 37°C. In the control groups, Lec-2 cells were untreated or incubated with either sialyltransferase or CMP-sialic acid. The six cell lines utilized in the present study were obtained from the American Type Culture Collection (Manassas, VA) and maintained at 37°C with 5% CO2 in their respective media, supplemented with 10% fetal bovine serum and penicillin-streptomycin. N-acetylneuraminic acid is the most widespread sugar of the sialic acid family whose members are frequently found as a terminal sugar in cell surface complex carbohydrates and are known to play a major role in many processes of biological recognition such as cellular adhesion and binding of toxins and virus (Varki, 1993). All sialic acids are biosynthetically derived from Neu5Ac by the introduction of various modifications such as methylation, acetylation or sulfation.


